Organic
Functional Groups:
Aldehydes, ketones, 1° alcohols, etc.
Organic chemistry is dominated by the "functional group approach", where organic molecules are deemed to be constructed from: An inert hydrocarbon skeleton onto which functional groups (FGs) are attached or superimposed.
The functional group approach "works" because the properties and reaction chemistry of a particular functional group (FG) can be remarkably independent of environment.
Therefore, it is only necessary to know about the chemistry of a few generic functions in order to predict the chemical behaviour of thousands of real organic chemicals.
Organic molecules are also named using the functional group approach:
2-hexanone
2-hexanol
2-chlorohexane
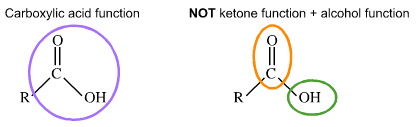
The rule is that functions assume their distinct identity when separated by –CH2– groups. Thus, the carbonyl, C=O, and hydroxy, OH, of a carboxylic acid, RCOOH, are part of a single function and are NOT "alcohol-plus-ketone":
A Couple of Words About The Functional Group Approach
The functional group approach is 100% empirical in that it is determined by experiment and experience, and not by theory (unlike VSEPR, for example.)
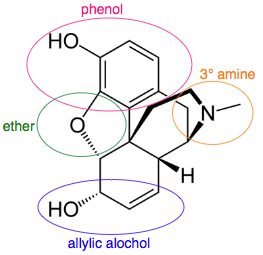
A multifunctional entity like the drug molecule morphine has several functional groups and chiral centres:
Professional chemists consider large multifunctional organic molecules in terms of 'substructures' rather than functional groups. Ring systems, for example, are better considered as substructures, although the dividing line can be fuzzy...
What You Need To Know
To be proficient in organic chemistry at university entrance level [ie, American AP, British A-Level or French Baccalaureate] exam systems,
in other words be able to:
name organic molecules
predict solubility in different types of solvent
predict chemical reactivity
predict spectra
it is absolutely essential to be able to recognise the 30 or so functions listed below. For a more comprehensive list of organic FGs, try the Chemogenesis Web Book FG Database:
| Alkane | Alkyl, and occasionally aryl (aromatic) functions are represented by the R–
Methyl: CH3– Ethyl: CH3CH2– Propyl: CH3CH2CH2– Isopropyl: (CH3)2CH– Phenyl: C6H5– etc. |
| Alkyl halide | Alkyl halides [haloalkanes] consist of an alkyl group attached to a halogen: F, Cl, Br, I. Chloro, bromo and iodo alkyl halides are often susceptible to elimination and/or nucleophilic substitution reactions. |
| Primary alcohol | Primary alcohols have an -OH function attached to an R-CH2- group. Primary alcohols can be oxidised to aldehydes and on to carboxylic acids. (It can be difficult to stop the oxidation at the aldehyde stage.) Primary alcohols can be shown in text as: RCH2OH |
| Secondary alcohol | Secondary alcohols have an -OH function attached to a R2CH- group. Secondary alcohols can be oxidised to ketones. Secondary alcohols can be shown in text as: R2CHOH |
| Tertiary alcohol | Tertiary alcohols have an -OH function attached to a R3C- group. Tertiary alcohols are resistant to oxidation with acidified potassium dichromate(VI), K. Tertiary alcohols can be shown in text as: R3COH |
| Aldehyde | Aldehydes have a hydrogen and an alkyl (or aromatic) group attached to a carbonyl function. Aldehydes can be shown in text as: RCHO Aldehydes are easily oxidised to carboxylic acids, and they can be reduced to primary alcohols. Aldehydes can be distinguished from ketones by giving positive test results with Fehlings solution (brick red precipitate) or Tollens reagent (silver mirror). Aldehydes give red-orange precipitates with 2,4-dinitrophenyl hydrazine. |
| Ketone | Ketones have a pair of alkyl or aromatic groups attached to a carbonyl function. Ketones can be shown in text as: RCOR Ketones can be distinguished from aldehydes by giving negative test results with Fehling?s solution (brick red precipitate) or Tollens reagent (silver mirror). Ketones give red-orange precipitates with 2,4-dinitrophenyl hydrazine. |
| Carboxylic acid | Carboxylic acids have an alkyl or aromatic groups attached to a hydroxy-carbonyl function. Carboxylic acids can be shown in text as: RCOOH Carboxylic acids are weak Bronsted acids and they liberate CO2 from carbonates and hydrogen carbonates. |
| Carbonyl function | The carbonyl group is a super function because many common functional groups are based on a carbonyl, including: aldehydes, ketones, carboxylic acids, esters, amides, acyl (acid) chlorides, acid anhydrides |
| Ester | Esters have a pair of alkyl or aromatic groups attached to a carbonyl + linking oxygen function. Esters can be shown in text as: RCOOR or (occasionally) ROCOR. carboxylic acid + alcohol -> ester + water This is an acid catalysed equilibrium. |
| Amide | Primary amides (shown) have an alkyl or aromatic group attached to an amino-carbonyl function. Primary amides can be shown in text as: RCONH2 Secondary amides have an alkyl or aryl group attached to the nitrogen: RCONHR Tertiary amides have two alkyl or aryl group attached to the nitrogen: RCONR2 |
| Primary amine | Primary amines have an alkyl or aromatic group and two hydrogens attached to a nitrogen atom. Primary amines can be shown in text as: RNH2 Primary amines are basic functions that can be protonated to the corresponding ammonium ion. Primary amines are also nucleophilic. |
| Secondary amine | Secondary amines have a pair of alkyl or aromatic groups, and a hydrogen, attached to a nitrogen atom. Secondary amines can be shown in text as: R2NH Secondary amines are basic functions that can be protonated to the corresponding ammonium ion. Secondary amines are also nucleophilic. |
| Tertiary amine | Tertiary amines have three alkyl or aromatic groups attached to a nitrogen atom. Tertiary amines can be shown in text as: R3N Tertiary amines are basic functions that can be protonated to the corresponding ammonium ion. Tertiary amines are also nucleophilic. |
| Acid chloride | Acid chlorides, or acyl chlorides, have an alkyl (or aromatic) group attached to a carbonyl function plus a labile (easily displaced) chlorine. Acid chlorides highly reactive entities are highly susceptible to attack by nucleophiles. Acid chlorides can be shown in text as: R-COCl or RCOCl |
| Acid anhydride | Acid anhydrides are formed when water is removed from a carboxylic acid, hence the name. Acid anhydrides can be shown in text as: (RCO)2O |
| Nitrile | Nitriles (or organo cyanides) have an alkyl (or aromatic) group attached to a carbon-triple-bond-nitrogen function. Nitriles can be shown in text as: RCN Note that there is a nomenclature issue with nitriles/cyanides. If a compound is named as the nitrile then the nitrile carbon is counted and included, but when the compound is named as the cyanide it is not. For example: CH3CH2CN is called propane nitrile or ethyl cyanide (cyanoethane). |
| Carboxylate ion or salt | Carboxylate ions are the conjugate bases of carboxylic acids, ie. the deprotonated carboxylic acid. Carboxylate ions can be shown in text as: RCOO? When the counter ion is included, the salt is being shown. Salts can be shown in text as: RCOONa |
| Ammonium ion | Ammonium ions have a total of four alkyl and/or hydrogen functions attached to a nitrogen atom.
[NH4]+ [RNH3]+ [R2NH2]+ [R3NH]+ [R4N]+ Quaternary ammonium ions are not proton donors, but the others are weak Bronsted acids (pKa about 10). |
| Amino acid | Amino acids, strictly alpha-amino acids, have carboxylic acid, amino function and a hydrogen attached to a the same carbon atom. There are 20 naturally occurring amino acids. All except glycine (R = H) are chiral and only the L enantiomer is found in nature. Amino acids can be shown in text as: R-CH(NH2)COOH |
| Alkene | Alkenes consist of a C=C double bond function.
Alkenes can be shown in text as:
Mono substituted: RCH=CH2 1,1-disubstituted: R2C=CH2 1,2-disubstituted: RCH=CHR Alkenes are planar and there is no rotation about the C=C bond. Alkenes are electron rich reactive centres and are susceptible to electrophilic addition. |
| trans-Alkene | trans-alkenes are 1,2-disubstituted functions with the two R, X or other groups on opposite sides of the C=C function. Due to the non-rotation of the C=C bond, cis and trans geometric isomers are not [thermally] Interconverted. |
| cis-Alkene | cis-Alkenes are 1,2-disubstituted functions with the two R, X or other groups on the same side of the C=C function. Due to the non-rotation of the C=C bond, cis and trans geometric isomers are not [thermally] Interconvertion. |
| Ether | Ethers have a pair of alkyl or aromatic groups attached to a linking oxygen atom. Ethers can be shown in text as: ROR Ethers are surprisingly unreactive and are very useful as solvents for many many (but not all) classes of reaction. |
| Alkoxide ion | Alkoxide ions an alkyl group attached to an oxyanion. Alkoxide ions can be shown in text as: RO? Sodium alkoxides, RONa, are slightly stronger bases than water and so cannot be prepared in water. Instead they are prepared by adding sodium to the dry alcohol. |
| Hydroxynitrile | Hydroxynitriles (also called cyanohydrins) are formed when hydrogen cyanide, H+ CN?, adds across the carbonyl function of an aldehyde or ketone. |
| Primary carbocation | Primary carbocations have a single alkyl function attached to a carbon centre with a formal positive charge. Carbocations - also and more correctly called carbenium ions - are important reactive intermediates implicated in electrophilic addition reactions and electrophilic aromatic substitution reactions. Stability: primary << secondary << tertiary |
| Secondary carbocation | Secondary carbocations have a pair of alkyl functions attached to a carbon centre with a formal positive charge. Carbocations - also and more correctly called carbenium ions - are important reactive intermediates implicated in electrophilic addition reactions and electrophilic aromatic substitution reactions. Stability: primary << secondary << tertiary |
| Tertiary carbocation | Tertiary carbocations have three alkyl functions attached to a carbon centre with a formal positive charge. Carbocations - also and more correctly called carbenium ions - are important reactive intermediates implicated in electrophilic addition reactions and electrophilic aromatic substitution reactions. Stability: primary << secondary << tertiary |
| Acyl cation | Acyl cations have an alkyl (or aromatic) group attached to a carbonyl function with a formal positive charge. Acyl cations are important reactive intermediates and are implicated in electrophilic addition reactions and electrophilic aromatic substitution reactions. Acyl cations are commonly formed from the corresponding acyl/acid chloride plus aluminium chloride. |
| Polymer | Polymers consist of small monomer molecules that have reacted together so as to form a large covalently bonded structure. There are two general types of polymerisation: addition and condensation. Linear chain polymers are generally thermoplastic, while three dimensional network polymers are not. |
| Diol or polyol | Diols and polyols are alcohols with two or more -OH functions. Diols and polyols are very soluble in water. They are used as high temperature polar solvents. |
(Indonesian & Polish Translations of this page)
© Mark R. Leach 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com
This free, open access web book is an ongoing project and your input is appreciated.